Water SARS-CoV-2 RT-PCR Test
Detect and quantify SARS-CoV-2 in wastewater
Colilert

Detect or quantify total coliforms and Escherichia coli, with results in 24 hours
Path-Chek Hygiene Tests

Rapid tests for the detection of a range of bacteria from work surfaces and the processing environment.
The range of Path-Chek Hygiene Systems consist of tests for 3 organism types; Listeria, Coliforms and Salmonella.
Biochemical Identification Kits
A range of simple, reliable bacterial identification systems for clinical, food, environmental and pharmaceutical laboratories.
The Microgen range of biochemical identification systems consists of: Microgen GN-ID, Microgen Listeria-ID, Microgen Bacillus-ID, Microgen Staph-ID, Microgen Strep-ID
Latex Agglutination Kits

Simple one-step identification and confirmation of a range of pathogenic bacteria. The benefits of these products include: Speed – Simplicity – Specificity – Cost
Available tests for: Salmonella spp., Campylobacter spp., C.difficile, Staphylococcus aureus, Streptococcal Grouping, Legionella pneumophila, E.coli O157, Listeria spp.
Captivate™
Captivate™ is a range of antibody coated paramagnetic particles for the specific immunomagnetic separation (IMS) of microorganisms. Purified antibodies to surface components of the target microorganism are covalently coupled to paramagnetic particles. Careful antibody selection produces a highly specific system designed for microbiological immunomagnetic separation of target bacteria from enrichment culture. IMS increases the sensitivity of the methodology and, in most circumstances, results are achieved 24 hours earlier than using standard protocols. Offered as part of a standard range and as bespoke products, Captivate bacterial immunomagnetic separation can also serve as a capture system for rapid detection systems.
Supplements & Additives
We offer an extensive range of media supplements and additives to complement our dehydrated culture media product lines. Supplements and additives can be used to assist in the isolation, selection, enrichment or growth of micro-organisms.
Dehydrated Culture Media

Dehydrated microbiological culture media are fundamental to the day-to-day operation of most microbiology laboratories, delivering numerous benefits.
Convenient – use straight from the shelf – little need for refrigeration or specialist storage
Waste-free – make up only what you need, when you need it
Long shelf-life – years compared with days/weeks for ready prepared media
Specialist – the easiest way to add and control sensitive, non-autoclavable components
Cost-effective – fits all lab regimes and work loads
Harlequin™ Chromogenic Media
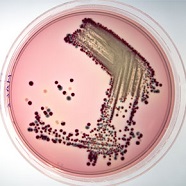
Chromogenic culture media provide a rapid and accurate method of isolating and enumerating target micro-organisms based on the detection of specific enzyme activities. Not only do chromogenic media enable faster detection of specific micro-organisms compared with classical culture media, they also improve sensitivity and can reduce the need for subculture or confirmatory tests. Lab M’s Harlequin range of chromogenic media includes products for the isolation of Listeria species, Salmonellae, Escherichia coli and Enterobacter (formerly Cronobacter) sakazakii.
Celsis Accel® system

takes the robustness and ease of use of the widely
adopted Celsis Advance II™ platform and packages
it into a small footprint. With a throughput of 30 assays
per hour, this system is specifically designed for small
and mid-sized pharmaceutical, home,
and beauty product manufacturing facilities.
Celsis Advance II™ system

is designed to deliver definitive results quickly.
Capable of running 120 assays per hour, high throughput
manufacturers can cut days off their production cycles and
reduce working capital requirements.
LAL-Accessories
Charles River provides all the necessary endotoxin-free test tubes and accessory products required to run an LAL assay. All accessory products are provided with a certificate of quality certifying appropriate endotoxin levels. These high-quality accessories impart control and consistency to LAL assays and the LAL laboratory, thereby minimizing invalid results and repeat testing.
